
Chemistry, 25.07.2020 03:01 jessica112776
Use the balanced combustion reaction above to calculate the enthalpy of combustion for C8H16. C8H16(1)= -174.5kJ/mol. I have no clue how to start this question and need help including the formulas so I know how to do it and some step by step commentary.


Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 14:20
Which of the following are sources of revenue for media companies? a. direct sales to producers b.advertising and subscriptions c. online purchase d. capital investments
Answers: 1

Chemistry, 22.06.2019 15:30
Draw the lewis dot structure for each of the following polyatomic ions
Answers: 1

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3
You know the right answer?
Use the balanced combustion reaction above to calculate the enthalpy of combustion for C8H16. C8H16(...
Questions


Mathematics, 02.09.2021 19:40


Mathematics, 02.09.2021 19:40

Geography, 02.09.2021 19:40




English, 02.09.2021 19:40

English, 02.09.2021 19:40

Mathematics, 02.09.2021 19:40


Mathematics, 02.09.2021 19:40

Mathematics, 02.09.2021 19:40



Chemistry, 02.09.2021 19:40



History, 02.09.2021 19:50

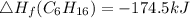
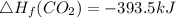
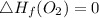
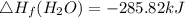
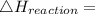 8 x - 393.5 - 8 x 285.82 + 174.5x 1
8 x - 393.5 - 8 x 285.82 + 174.5x 1 

