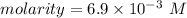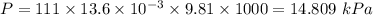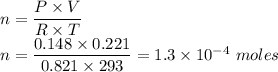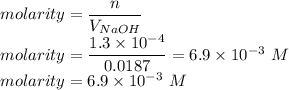Chemistry, 24.07.2020 17:01 taylorclarkx17
The volume of a sample of pure HCl gas was 221 mL at 20°C and 111 mmHg. It was completely dissolved in about 50 mL of water and titrated with an NaOH solution; 18.7 mL of the NaOH solution was required to neutralize the HCl. Calculate the molarity of the NaOH solution.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:00
Which of the following is the correct formula for copper (i) sulfate trihydrate? cuso4 · 3h2o cuso4(h2o)3 cu2so4(h2o)3 cu2so4 · 3h2o
Answers: 1


Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2

Chemistry, 23.06.2019 01:00
Which statement is true regarding the diagram of circle p? the sum of y and z must be 2x. the sum of y and z must be x. the difference of z and y must be 2x. the difference of z and y must be x
Answers: 1
You know the right answer?
The volume of a sample of pure HCl gas was 221 mL at 20°C and 111 mmHg. It was completely dissolved...
Questions

Computers and Technology, 18.07.2019 10:20



Chemistry, 18.07.2019 10:20

Chemistry, 18.07.2019 10:20


Chemistry, 18.07.2019 10:20

Biology, 18.07.2019 10:20





Business, 18.07.2019 10:20

Social Studies, 18.07.2019 10:20




English, 18.07.2019 10:20


Computers and Technology, 18.07.2019 10:20