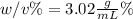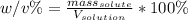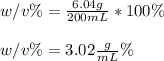An aqueous solution of cobalt(II) fluoride, , is made by dissolving 6.04 grams of cobalt(II) fluoride in sufficient water in a 200. mL volumetric flask, and then adding enough water to fill the flask to the mark. What is the weight/volume percentage of cobalt(II) fluoride in the solution

Answers: 1
Another question on Chemistry


Chemistry, 21.06.2019 21:30
Fugu, also known as puffer fish, is a sushi delicacy that can also be lethal. puffer fish contain a powerful toxin that can kill an adult a few hours after ingestion. sushi chefs who prepare fugu must be specially trained because any contamination of the toxin-free areas of the fish can be deadly. recently this toxin has been put to good use, as scientists have discovered that a purified form of it can treat severe pain in cancer patients. this recent scientific discovery would fall under which area of chemistry? applied biochemistry pure organic chemistry pure physical chemistry applied inorganic chemistry
Answers: 1

Chemistry, 22.06.2019 06:00
Calculate - analysis of compound composed of iron and oxygen yields 174.86 of fe and 75.14g of o. what is the empirical formula for this compound?
Answers: 3

Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1
You know the right answer?
An aqueous solution of cobalt(II) fluoride, , is made by dissolving 6.04 grams of cobalt(II) fluorid...
Questions

History, 09.02.2021 22:30


Arts, 09.02.2021 22:30


Mathematics, 09.02.2021 22:30

Mathematics, 09.02.2021 22:30


History, 09.02.2021 22:30

Mathematics, 09.02.2021 22:30



Mathematics, 09.02.2021 22:30

Mathematics, 09.02.2021 22:30

Mathematics, 09.02.2021 22:30

Medicine, 09.02.2021 22:30

Health, 09.02.2021 22:30




Mathematics, 09.02.2021 22:30