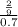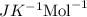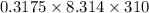Chemistry, 22.07.2020 23:01 MidnightAIY179
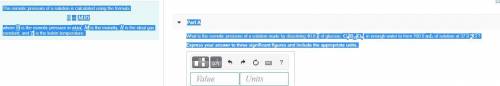
The osmotic pressure of a solution is calculated using the formula Π=MRT where Π is the osmotic pressure in atm, M is the molarity, R is the ideal gas constant, and T is the kelvin temperature. Part A What is the osmotic pressure of a solution made by dissolving 40.0 g of glucose, C6H12O6, in enough water to form 700.0 mL of solution at 37.0 ∘C ? Express your answer to three significant figures and include the appropriate units. nothing nothing

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 23.06.2019 01:30
Ascientist is measuring the pressure that is exerted by each of the following gases in the atmosphere: carbon dioxide, oxygen, and nitrogen. which term most likely describes what she is measuring?
Answers: 1

Chemistry, 23.06.2019 05:40
Why is any chemical reaction always balanced? give reasons and explain the easiest way to solve the balancing problems in chemical equations with stoichiometric coefficients upto 20 as hit and trial doesn't always work. give full reasoning
Answers: 1
You know the right answer?
The osmotic pressure of a solution is calculated using the formula Π=MRT where Π is the osmotic pres...
Questions

Geography, 16.02.2021 22:50


English, 16.02.2021 22:50






Chemistry, 16.02.2021 22:50




Biology, 16.02.2021 22:50


Mathematics, 16.02.2021 22:50



Mathematics, 16.02.2021 22:50


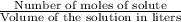
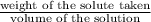
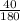
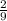 moles
moles