Chemistry, 19.07.2020 14:01 sonnyboi2305
Given the following thermochemical data C(s) + ½O2(g) ---> CO(g) ∆H1 = -111 kJ/mol C(s) + O2(g) ---> CO2(g) ∆H2 = -395 kJ/mol What is the ∆Hr for the reaction: CO(g) + ½O2(g) ---> CO2(g) ∆Hr = ?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 16:00
Which statement describes the appearance of a temperature-vs.-time graph? a horizontal line shows that the temperature increases at a constant rate over time. a vertical line shows that the temperature decreases at a constant rate over time. horizontal lines where the temperature is constant during phase changes connect upward-sloping lines where the temperature increases. horizontal lines where the temperature increases are connected by upward-sloping lines where the temperature is constant for each phase.
Answers: 1


Chemistry, 22.06.2019 23:30
What are the similarities between compounds and mixtures?
Answers: 3

Chemistry, 23.06.2019 01:00
If a straight-chain hydrocarbon is a gas at room temperature, how many carbon atoms will it have? a. 6 carbon atoms b. 12 carbon atoms c. 24 carbon atoms d. 3 carbon atoms
Answers: 1
You know the right answer?
Given the following thermochemical data C(s) + ½O2(g) ---> CO(g) ∆H1 = -111 kJ/mol C(s) + O2(g) -...
Questions


English, 13.10.2019 14:50



Health, 13.10.2019 14:50



Mathematics, 13.10.2019 14:50


Physics, 13.10.2019 14:50



Mathematics, 13.10.2019 14:50

History, 13.10.2019 14:50



History, 13.10.2019 14:50


English, 13.10.2019 14:50

Mathematics, 13.10.2019 14:50

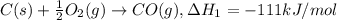 ..[1]
..[1]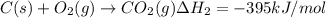 ...[2]
...[2]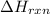 of following reaction :
of following reaction :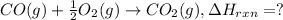 ..[3]
..[3]