
1. A 10.0 g sample of propane, C3H8, was combusted in a constant-volume bomb
calorimeter. The total heat capacity of the bomb calorimeter and water was 8.0
kJ/°C. The molar heat of combustion of propane is -2 222 KJ/mol. If the starting
temperature of the water was 20 °C, what will be the final temperature of the
bomb calorimeter?

Answers: 3
Another question on Chemistry

Chemistry, 23.06.2019 03:30
Ineed pls urgent 1-20 in order and fully detail step my step.
Answers: 1

Chemistry, 23.06.2019 05:30
The term gas is limited to those substances that exist in the gaseous state at
Answers: 1

Chemistry, 23.06.2019 16:00
What is a three-dimensional structure that represents the alternating pattern of particles in a crystal? a polyatomic ion a crystal lattice an ionic bond a formula unit
Answers: 3

Chemistry, 23.06.2019 20:30
Atmospheric pressure is 1.00 atm, but air is only composed of 21.0% (by moles) oxygen. thus, the partial pressure of oxygen in air is less than 1.00 atm. what is the millimolar concentration of dissolved oxygen for a lake or stream under the partial pressure of oxygen and a temperature of 20 ∘c?
Answers: 1
You know the right answer?
1. A 10.0 g sample of propane, C3H8, was combusted in a constant-volume bomb
calorimeter. The total...
Questions


Advanced Placement (AP), 22.10.2020 01:01

Mathematics, 22.10.2020 01:01


Mathematics, 22.10.2020 01:01



Mathematics, 22.10.2020 01:01



Mathematics, 22.10.2020 01:01



Mathematics, 22.10.2020 01:01


Engineering, 22.10.2020 01:01


Mathematics, 22.10.2020 01:01

English, 22.10.2020 01:01

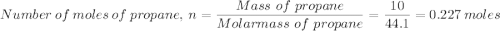 Which gives;
Which gives; in the calorimeter = Heat capacity × Temperature change
in the calorimeter = Heat capacity × Temperature change

