Chemistry, 20.07.2020 01:01 cheesecake1919
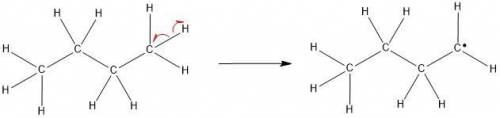
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG (Liquefied Petroleum Gas) cylinders (a common source of cooking gas). It has two arrangements of carbon atoms: a straight chain and a branched chain. Using this information, draw the structure of the tertiary butyl radical that will form upon removal of a hydrogen atom.
Requried:
Draw the molecule on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars. Include all free radicals by right-clicking on an atom on the canvas and then using the Atom properties to select the monovalent radical.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 09:30
What are scientists who study fossils called? ( a ) astronomers. ( b ) biologists. ( c ) geologists. ( d ) paleontologists.
Answers: 2

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1


Chemistry, 22.06.2019 23:10
Amines are good nucleophiles, even though they are neutral molecules. how would the rate of an sn2 reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? amines are good nucleophiles, even though they are neutral molecules. how would the rate of an reaction between an amine and an alkyl halide be affected if the polarity of the solvent is increased? because both reactants in the rate-limiting step are neutral, the reaction will be faster if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will be slower if the polarity of the solvent is increased. because both reactants in the rate-limiting step are neutral, the reaction will occur at the same rate if the polarity of the solvent is increased. request answer
Answers: 3
You know the right answer?
The molecular formula of butane is C4H10. It is obtained from petroleum and is used commonly in LPG...
Questions


Mathematics, 14.02.2020 18:30


Mathematics, 14.02.2020 18:30

Mathematics, 14.02.2020 18:30



Arts, 14.02.2020 18:30


Spanish, 14.02.2020 18:30



History, 14.02.2020 18:30

Social Studies, 14.02.2020 18:31





Geography, 14.02.2020 18:31