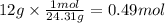Chemistry, 15.07.2020 02:01 ivanyeli4520
How many moles of hydrogen gas will be produced when 12 g of Mg will react completely with excess of an acid according to the following reaction? 2 Mg + 2 HCI — 2 MgCl2 + H,

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:30
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 10:50
Someone offer some answers to this, i will give 98 coins and mark as brainliest! i will put the rest of the lab down in the comments,solutions pre-lab questions: in this lab, you will make fruit drinks with powdered drink mix. complete the pre-lab questions to get the values you need for your drink solutions. calculate the molar mass of powered fruit drink mix, made from sucrose (c12h22o11).using stoichiometry, determine the mass of powdered drink mix needed to make a 1.0 m solution of 100 ml. (hint: use molarity = to find the moles of drink mix, then convert moles to grams using a mole conversion.)what mass of powdered drink mix is needed to make a 0.5 m solution of 100 ml?
Answers: 1

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
How many moles of hydrogen gas will be produced when 12 g of Mg will react completely with excess of...
Questions


History, 25.07.2019 19:30




World Languages, 25.07.2019 19:30



Business, 25.07.2019 19:30



Biology, 25.07.2019 19:30

History, 25.07.2019 19:30


Chemistry, 25.07.2019 19:30

Chemistry, 25.07.2019 19:30

Social Studies, 25.07.2019 19:30

History, 25.07.2019 19:30