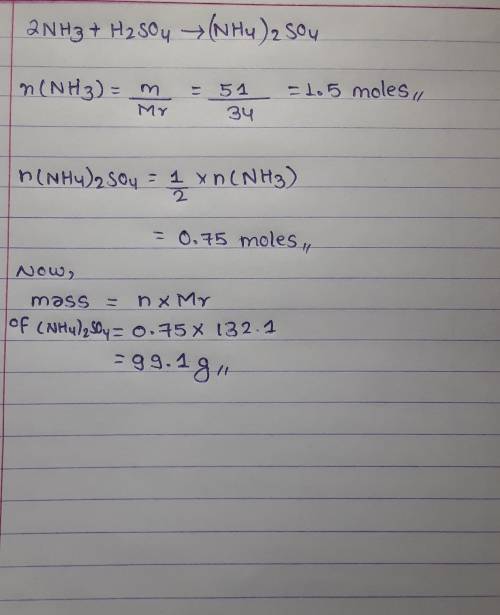Chemistry, 14.07.2020 01:01 saraaaaaaaa20
calculate the mass of ammonium sulfate that can be made from 51 grams of ammonia. 2NH₃ + H₂SO₄ → (NH₄)₂SO₄ (keeping the mole concept in mind)

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Can you reduce your impairment by drinking coffee or cold showers? true or false?
Answers: 3

Chemistry, 21.06.2019 15:00
Which element in the third period would you expect to have the larger atomic radius, sodium (na) or sulfur (s)? a. sodium, because it has a higher effective nuclear charge attracting electrons in fewer energy levels. b. sodium, because it has fewer protons attracting electrons in the same energy levels. c. sulfur, because it has more protons attracting electrons in more energy levels. d. sulfur, because it has a higher effective nuclear charge attracting electrons in the same energy levels.
Answers: 2

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

You know the right answer?
calculate the mass of ammonium sulfate that can be made from 51 grams of ammonia. 2NH₃ + H₂SO₄ → (NH...
Questions



Business, 03.12.2020 14:00





Chemistry, 03.12.2020 14:00


Arts, 03.12.2020 14:00

Biology, 03.12.2020 14:00

Business, 03.12.2020 14:00



Mathematics, 03.12.2020 14:00

Physics, 03.12.2020 14:00

English, 03.12.2020 14:00

History, 03.12.2020 14:00

History, 03.12.2020 14:00