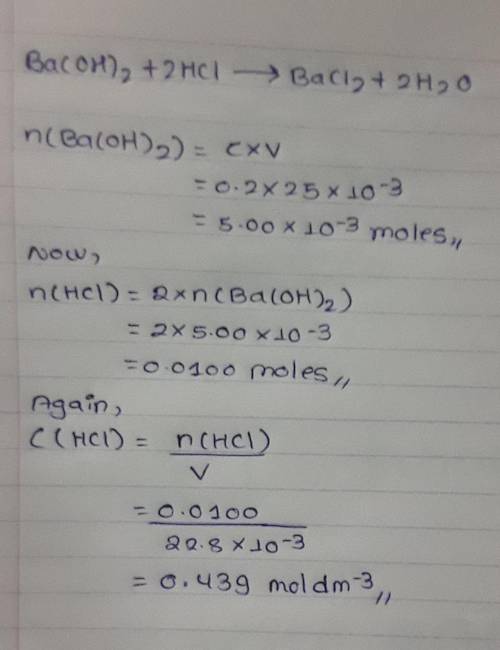Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 23.06.2019 14:00
How does electronegativity changes as we move from left to right across a period
Answers: 2

Chemistry, 23.06.2019 21:30
Fertilizer is sold in 100 pound bags labelled with the amount of nitrogen ( n), phosphoric acid ( p2o5), and potash ( k2o) present. the mixture of these nutrients varies from one type of fertilizer to the next. for example, a 100100 pound bag of vigoro ultra turf fertilizer contains 2929 pounds of nitrogen, 33 pounds of phosphoric acid, and 44 pounds of potash. another type of fertilizer, parker's premium starter, has 1818 pounds of nitrogen, 2525 pounds of phosphoric acid, and 66 pounds of potash per 100100 pounds. determine the amount of each type required to yield a mixture containing the 112112 pounds of nitrogen, 8181 pounds of phosphoric acid, and 2626 pounds of potash.
Answers: 3
You know the right answer?
25cm3 of 0.2mol/dm3 barium hydroxide solution reacted with 22.8cm3 hydrochloric acid. Calculate the...
Questions

Social Studies, 27.05.2021 16:30


History, 27.05.2021 16:30


Mathematics, 27.05.2021 16:30

Computers and Technology, 27.05.2021 16:30

Mathematics, 27.05.2021 16:30

History, 27.05.2021 16:30




Mathematics, 27.05.2021 16:40


Mathematics, 27.05.2021 16:40

History, 27.05.2021 16:40

Mathematics, 27.05.2021 16:40


Health, 27.05.2021 16:40