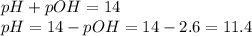Chemistry, 15.07.2020 01:01 29delphina
What is the pH of a 0.300 M NH₃ solution that has Kb = 1.8 × 10⁻⁵ ? The equation for the dissociation of NH₃ is: NH₃ (aq) + H₂O (l) ⇄ NH₄⁺ (aq) + OH⁻ (aq)

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Use the drop-down menus to answer each question. which runner finished the 100 m race in the least amount of time? which runner stopped running for a few seconds during the race? at what distance did anastasia overtake chloe in the race?
Answers: 1

Chemistry, 22.06.2019 05:00
Cucl2 + 2nano3 cu(no3)2 + 2nacl what is the percent yield of nacl if 31.0 g of cucl2 reacts with excess nano3 to produce 21.2 g of nacl? 49.7% 58.4% 63.6% 78.7%
Answers: 1

Chemistry, 22.06.2019 07:00
In the cathode ray tube experiment, j. j. thomson passed an electric current through different gases inside a cathode ray tube in the presence of an electric field. in which two ways did this experiment change scientists’ understanding of the atom?
Answers: 2

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1
You know the right answer?
What is the pH of a 0.300 M NH₃ solution that has Kb = 1.8 × 10⁻⁵ ? The equation for the dissociatio...
Questions



Mathematics, 16.05.2020 12:57


Mathematics, 16.05.2020 12:57

English, 16.05.2020 12:57

Biology, 16.05.2020 12:57

English, 16.05.2020 12:57

Health, 16.05.2020 12:57


History, 16.05.2020 12:57

Mathematics, 16.05.2020 12:57

Chemistry, 16.05.2020 12:57

Mathematics, 16.05.2020 12:57



Mathematics, 16.05.2020 12:57


Mathematics, 16.05.2020 12:57

Arts, 16.05.2020 12:57

![[OH^{-} ]=\sqrt{Kb \times Cb } = \sqrt{1.8 \times 10^{-5} \times 0.300 } = 2.3 \times 10^{-3} M](/tpl/images/0706/3859/9462d.png)
![pOH =-log[OH^{-} ]= -log(2.3 \times 10^{-3} M) = 2.6](/tpl/images/0706/3859/e37b1.png)