
Chemistry, 14.07.2020 01:01 ondreabyes225pcr83r
Consider the following reaction where Kc = 6.50×10-3 at 298 K: 2NOBr(g) 2NO(g) + Br2(g) A reaction mixture was found to contain 9.83×10-2 moles of NOBr(g), 5.44×10-2 moles of NO(g), and 4.13×10-2 moles of Br2(g), in a 1.00 liter container. Is the reaction at equilibrium? If not, what direction must it run in order to reach equilibrium? The reaction quotient, Qc

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 04:50
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 08:00
What are the similarities of physical and chemical change ?
Answers: 1

Chemistry, 22.06.2019 14:00
Displacement is the slope of a velocity vs. time graph a. true b. false
Answers: 1
You know the right answer?
Consider the following reaction where Kc = 6.50×10-3 at 298 K: 2NOBr(g) 2NO(g) + Br2(g) A reaction m...
Questions













Social Studies, 13.12.2019 06:31


English, 13.12.2019 06:31



Mathematics, 13.12.2019 06:31


Chemistry, 13.12.2019 06:31

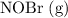 ) in order to reach an equilibrium.
) in order to reach an equilibrium.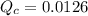 .
.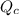 is equal to the equilibrium constant
is equal to the equilibrium constant 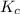 .
. 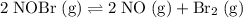 .
.![[\mathrm{NOBr\; (g)}]](/tpl/images/0704/6105/9e238.png) ,
, ![[\mathrm{NO\; (g)}]](/tpl/images/0704/6105/b4859.png) , and
, and ![[\mathrm{Br_2\; (g)}]](/tpl/images/0704/6105/fc290.png) denote the concentration of the three species. The formula for the reaction quotient of this system will be:
denote the concentration of the three species. The formula for the reaction quotient of this system will be:![\displaystyle Q_c = \frac{[\mathrm{NO\; (g)}]^2 \cdot [\mathrm{Br_2\; (g)}]}{[\mathrm{NOBr\; (g)}]^2}](/tpl/images/0704/6105/e21d0.png) .
.![\displaystyle Q_c = \frac{[\mathrm{NO\; (g)}]^2 \cdot [\mathrm{Br_2\; (g)}]}{[\mathrm{NOBr\; (g)}]^2} \approx 1.26\times 10^{-2} = 0.0126](/tpl/images/0704/6105/6c71a.png) .
.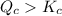 . Since
. Since 

