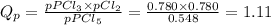Chemistry, 13.07.2020 21:01 ROBIOX5934
At T = 250 °C the reaction PCl5(g) PCl3(g) + Cl2(g) has an equilibrium constant in terms of pressures Kp = 2.15. (a) Suppose the initial partial pressure of PCl5 is 0.548 atm, and PPCl3 = PCl2 = 0.780 atm. Calculate the reaction quotient Qp and state whether the reaction proceeds to the right or to the left as equilibrium is approached

Answers: 1
Another question on Chemistry




Chemistry, 22.06.2019 20:00
I’m an electrically neutral atomic any element, there are equal numbers of
Answers: 2
You know the right answer?
At T = 250 °C the reaction PCl5(g) PCl3(g) + Cl2(g) has an equilibrium constant in terms of pressure...
Questions



Mathematics, 27.07.2019 07:00


Mathematics, 27.07.2019 07:00

English, 27.07.2019 07:00


Health, 27.07.2019 07:00







Physics, 27.07.2019 07:00


History, 27.07.2019 07:00

History, 27.07.2019 07:00

History, 27.07.2019 07:00

History, 27.07.2019 07:00