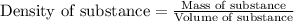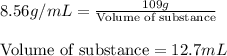Chemistry, 29.01.2020 14:03 LillianMRucker
you have a 100.0-ml graduated cylinder containing 50.0 ml of water. you carefully place a 109-g piece of brass (density = 8.56 g/ml) into the water. what is the final volume reading in the graduated cylinder?
show work !

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 23.06.2019 01:50
Ablock of aluminum is dropped into a graduated cylinder with an initial volume of water at 75ml and the volumes rises to 90ml. if the block has a mass of 40.5 g what is its density ?
Answers: 1

Chemistry, 23.06.2019 07:30
To separate a mixture of hard candies nd marbles the most efficient method would be
Answers: 3
You know the right answer?
you have a 100.0-ml graduated cylinder containing 50.0 ml of water. you carefully place a 109-g piec...
Questions

History, 17.04.2021 01:00






Mathematics, 17.04.2021 01:00







Mathematics, 17.04.2021 01:00


Mathematics, 17.04.2021 01:00


Mathematics, 17.04.2021 01:00

Mathematics, 17.04.2021 01:00

Arts, 17.04.2021 01:00