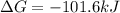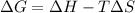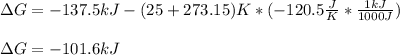Chemistry, 08.07.2020 20:01 michelle7511
Consider the following reaction: C2H4(g)+H2(g)→C2H6(g) ΔH=−137.5kJ; ΔS=−120.5J/K Calculate ΔG at 25 ∘C and determine whether the reaction is spontaneous. Express the free energy change in joules to four significant figures.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:30
Is a pencil falling to the floor anon contact force, a force, or a contact force
Answers: 1

Chemistry, 23.06.2019 00:30
In a ball-and-stick molecular model, what do the sticks represent?
Answers: 1

Chemistry, 23.06.2019 05:30
How many moles are in 1.26*10^24 particles in significant figures
Answers: 2

Chemistry, 23.06.2019 05:50
Which of the following isotopes has the same number of neutrons as phosphorus-31?
Answers: 1
You know the right answer?
Consider the following reaction: C2H4(g)+H2(g)→C2H6(g) ΔH=−137.5kJ; ΔS=−120.5J/K Calculate ΔG at 25...
Questions







Mathematics, 22.03.2021 21:00


Mathematics, 22.03.2021 21:00

English, 22.03.2021 21:00

Mathematics, 22.03.2021 21:00

Mathematics, 22.03.2021 21:00


Biology, 22.03.2021 21:00


English, 22.03.2021 21:00


Social Studies, 22.03.2021 21:00

Mathematics, 22.03.2021 21:00