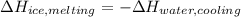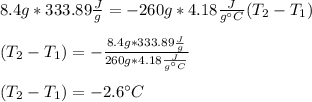An 8.4 g ice cube is placed into 260 g of water. Calculate the temperature change in the water upon the complete melting of the ice. Assume that all of the energy required to melt the ice comes from the water. Express your answer in terms of the initial temperature of water, T. a. -0.033T - 2.6 °Cb. -2.6T + 0.033 °Cc. 0.033T - 2.6 °Cd. 2.6T -0.033 °C

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
Idon't really understand this can you me and show your work.☺☺[ chemistry b] subject [ electron transfer in lonic bonds]grade( 12)
Answers: 1

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 16:00
Which process transfers heat from inside earth to its surface? convection currents in mantle pulling away of tectonic plates drawing in of tectonic plates convection currents in crust
Answers: 1

Chemistry, 22.06.2019 19:20
15. which of the following is not human-caused groundwater pollution? a. water in an aquifer dissolves elements such as arsenic and mercury from surrounding rock. b. water in an aquifer is contaminated by leachate that seeps into the ground from a landfill. c. water in an aquifer becomes polluted with chemicals used in hydraulic fracturing, or fracking. d. water in an aquifer absorbs harmful bacteria from the drainage field of a septic tank.
Answers: 1
You know the right answer?
An 8.4 g ice cube is placed into 260 g of water. Calculate the temperature change in the water upon...
Questions


Mathematics, 03.01.2021 23:50

Mathematics, 03.01.2021 23:50

Mathematics, 03.01.2021 23:50








Mathematics, 03.01.2021 23:50

Biology, 03.01.2021 23:50

Chemistry, 03.01.2021 23:50



Engineering, 03.01.2021 23:50

Mathematics, 03.01.2021 23:50

Advanced Placement (AP), 03.01.2021 23:50

Physics, 03.01.2021 23:50