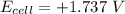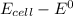Chemistry, 08.07.2020 02:01 gabbytumey
Consider an electrochemical cell based on the spontaneous reaction 2AgCl(s) + Zn(s) → 2Ag(s) + 2Cl– + Zn2+. If the zinc ion concentration is kept constant at 1 M, and the chlorine ion concentration is decreased from 1 M to 0.001 M, the cell voltage should:

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 15:40
What is the total reduction potential of a cell in which potassium (k) is reduced and copper (cu) is oxidized? a. 2.59 v b. 3.27 v c. -3.27 v d.-2.59 v
Answers: 1

Chemistry, 21.06.2019 22:30
Each of the following compounds contains a metal that can exhibit more than one ionic charge. provide systematic names for each of these compounds. (a) cr(clo3)6 (b) mo(cn)6 (c) cr2(so3)3 (d) v(clo2)2 (e) v(cn)5 (f) os(clo2)4
Answers: 3

Chemistry, 22.06.2019 06:00
Ethanol (c2h5oh) is produced from the fermentation of sucrose in the presence of enzymes. c12h22o11(aq) + h2o(g) 4 c2h5oh(l) + 4 co2(g) determine the theoretical yield and the percent yields of ethanol if 680. g sucrose undergoes fermentation and 326.5 g ethanol is obtained. theoretical _ g _ percent %
Answers: 1

Chemistry, 22.06.2019 15:30
How does a large body of water, such as the ocean, influence climate?
Answers: 1
You know the right answer?
Consider an electrochemical cell based on the spontaneous reaction 2AgCl(s) + Zn(s) → 2Ag(s) + 2Cl–...
Questions



Mathematics, 27.11.2019 02:31


History, 27.11.2019 02:31

English, 27.11.2019 02:31

Physics, 27.11.2019 02:31

History, 27.11.2019 02:31


Mathematics, 27.11.2019 02:31

Mathematics, 27.11.2019 02:31

Mathematics, 27.11.2019 02:31








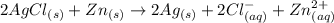
![E_{cell} = E^0- \dfrac{0,059}{n}log (\dfrac{[product]}{[reactant]})](/tpl/images/0702/8732/cb883.png)
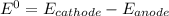
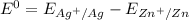
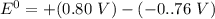
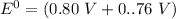
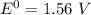
![E_{cell} = E^0- \dfrac{0.059}{n}log (\dfrac{[product]}{[reactant]})](/tpl/images/0702/8732/6f93c.png)
![E_{cell} = 1.56 - \dfrac{0.059}{2}log ({[Zn^{2+} ]}{[Cl^{2-}]})](/tpl/images/0702/8732/feb0e.png)
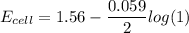
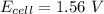
![E_{cell} = 1.56 - \dfrac{0.059}{2}log ({[1*0.001^2}]})](/tpl/images/0702/8732/27a68.png)
![E_{cell} = 1.56 - 0.0295 \ * \ log ({[1*10^{-6}}]})](/tpl/images/0702/8732/5785a.png)