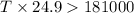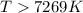For the reaction
N2(g) + O2(g)→2 NO(g) H° = 181 kJ and S° = 24.9 J/K G°
The equilibriu...

Chemistry, 08.07.2020 02:01 ordonez9029
For the reaction
N2(g) + O2(g)→2 NO(g) H° = 181 kJ and S° = 24.9 J/K G°
The equilibrium constant, K, would be greater than 1 at temperatures (above or below).. Kelvin.

Answers: 2
Another question on Chemistry

Chemistry, 20.06.2019 18:04
If this equation was completed which statement would it best support
Answers: 2


Chemistry, 22.06.2019 06:00
According to each substances heat of fusion, which of the items below requires more heat to be added per gram of substance to go from solid to liquid? silver sulfur water lead
Answers: 2

You know the right answer?
Questions

History, 03.08.2021 22:20



Mathematics, 03.08.2021 22:20


Chemistry, 03.08.2021 22:20



Mathematics, 03.08.2021 22:30

Computers and Technology, 03.08.2021 22:30

Mathematics, 03.08.2021 22:30

Mathematics, 03.08.2021 22:30


Mathematics, 03.08.2021 22:30

Advanced Placement (AP), 03.08.2021 22:30



History, 03.08.2021 22:30

English, 03.08.2021 22:30

Mathematics, 03.08.2021 22:30

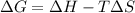
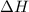 = enthalpy change = 181 kJ/mol = 181000 J/mol (1kJ=1000J)
= enthalpy change = 181 kJ/mol = 181000 J/mol (1kJ=1000J)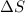 = entropy change= 24.9 J/Kmol
= entropy change= 24.9 J/Kmol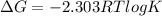
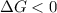
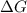 < 0,
< 0,  >
>