
Consider the intermediate chemical reactions. 2 equations. First: upper C a (s) plus upper C upper O subscript 2 (g) plus one half upper O subscript 2 (g) right arrow upper C a upper C upper O subscript 3 (s). Delta H 1 equals negative 812.8 kilojoules. Second: 2 upper C a (s) plus upper O subscript 2 (g) right arrow 2 upper C a upper O (s). Delta H 2 equals negative 1, 269 kilojoules. The final overall chemical equation is Upper Ca upper O (s) plus upper C upper O subscript 2 (g) right arrow upper C a upper C upper O subscript 3 (s).. When the enthalpy of this overall chemical equation is calculated, the enthalpy of the second intermediate equation is halved and has its sign changed. is halved. has its sign changed. is unchanged.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1


You know the right answer?
Consider the intermediate chemical reactions. 2 equations. First: upper C a (s) plus upper C upper O...
Questions




Mathematics, 17.12.2020 21:50


Biology, 17.12.2020 21:50

Biology, 17.12.2020 21:50

Biology, 17.12.2020 21:50


Mathematics, 17.12.2020 21:50

Arts, 17.12.2020 21:50



Mathematics, 17.12.2020 21:50

Mathematics, 17.12.2020 21:50



Mathematics, 17.12.2020 21:50

Mathematics, 17.12.2020 21:50

Mathematics, 17.12.2020 21:50

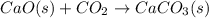
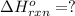
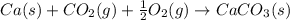
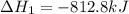
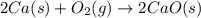
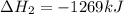
![\Delta H^o_{rxn}=[1\times (\Delta H_1)]+[\frac{1}{2}\times (-\Delta H_2)]](/tpl/images/0701/6838/89c09.png)


