Chemistry, 04.07.2020 23:01 WhiteMex69
408 J of energy is transferred to a system consisting of 2.0 moles of an ideal gas. If the volume of this gas stays at a constant 4.4 L, calculate the change in internal energy of the gas.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 13:00
Imagine that you push on a large rock. at what point does your effort change the rock’s mechanical energy?
Answers: 1


Chemistry, 23.06.2019 10:00
Which number should be placed before f2 on the reactants side equation to make equation balanced? xe + > xef4
Answers: 1

Chemistry, 23.06.2019 10:30
Ireally need ! calcium metal reacts with a potassium chloride solution to form calcium chloride and potassium ions. balance this reaction. (s) + (aq) → cacl2(s) + +(aq) a) 1, 2, 1, 2 b) 1, 2, 1, 1 c) 1, 1, 1, 1 d) 2, 1, 2, 1
Answers: 1
You know the right answer?
408 J of energy is transferred to a system consisting of 2.0 moles of an ideal gas. If the volume of...
Questions



Mathematics, 18.12.2020 19:40


Mathematics, 18.12.2020 19:40


Mathematics, 18.12.2020 19:40

Geography, 18.12.2020 19:40


Mathematics, 18.12.2020 19:40

Mathematics, 18.12.2020 19:40

Mathematics, 18.12.2020 19:40




Mathematics, 18.12.2020 19:40

Health, 18.12.2020 19:40


Mathematics, 18.12.2020 19:40

Mathematics, 18.12.2020 19:40

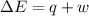
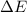 =Change in internal energy
=Change in internal energy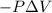
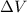 = 0 and w = 0.
= 0 and w = 0.