
Chemistry, 04.07.2020 23:01 uchapman6286
Ammonia (NH3) reacts with oxygen to form nitrogen monoxide and water. All the materials involved in this reaction are gasses. 0.100 moles of each of the reactants are initially introduced to a 5.0-liter reaction vessel. a. What would be the quantity of each gas in the container upon completion of the reaction? b. What would be the partial pressure of each gas on the reaction vessel upon reaction completion if the temperature of the system is 105 degrees C? c. What is the total pressure of all the gases on the reaction vessel at 105 degrees C?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
The balanced chemical equation for this lab is: 3cucl2(aq) + 2al(s) 3cu(s) + 2alcl3(aq) if 10.5 g copper chloride react with 12.4 g aluminum, what is the limiting reactant?
Answers: 3

Chemistry, 22.06.2019 03:30
The boiling point of liquids is very high what does it indicate
Answers: 1

Chemistry, 22.06.2019 07:20
2pos suppose an object in free fall is dropped from a building. its starting velocity is 0 m/s. ignoring the effects of air resistance, what is the speed (in m/s) of the object after falling 3 seconds? give your answer as a positive decimal without units. answer here
Answers: 1

Chemistry, 22.06.2019 12:00
What term is applied to a scientist who studies ancient life, including animal and plant fossils a. anthropologist b. dendroclimatologist c. geophysicist d. paleontologist
Answers: 2
You know the right answer?
Ammonia (NH3) reacts with oxygen to form nitrogen monoxide and water. All the materials involved in...
Questions

Mathematics, 14.11.2020 08:40

Mathematics, 14.11.2020 08:40

Mathematics, 14.11.2020 08:40


Mathematics, 14.11.2020 08:40


Mathematics, 14.11.2020 08:40

Mathematics, 14.11.2020 08:40

Mathematics, 14.11.2020 08:40




Mathematics, 14.11.2020 08:40

Chemistry, 14.11.2020 08:40

World Languages, 14.11.2020 08:40

History, 14.11.2020 08:40

Mathematics, 14.11.2020 08:40



Mathematics, 14.11.2020 08:40

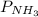 = 12,576.5 Pa,
= 12,576.5 Pa, 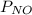 = 50,306.05 Pa,
= 50,306.05 Pa, 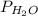 = 74,459.1 Pa
= 74,459.1 Pa

