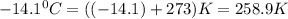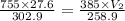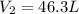Chemistry, 04.07.2020 14:01 aeshaalhemri
A weather balloon is inflated to a volume of 27.6 L at a pressure of 755 mmHg and a temperature of 29.9 ∘C. The balloon rises in the atmosphere to an altitude where the pressure is 385 mmHg and the temperature is -14.1 ∘C. Assuming the balloon can freely expand, calculate the volume of the balloon at this altitude.

Answers: 2
Another question on Chemistry

Chemistry, 23.06.2019 01:00
Aman applies a force of 500n to push a truck 100m down the street how much does he do?
Answers: 1

Chemistry, 23.06.2019 05:20
Explain how global warming could have affected yellowstone frog and salamander habitat's, resulting in changes in the populations of these species
Answers: 2

Chemistry, 23.06.2019 05:50
Which of the following isotopes has the same number of neutrons as phosphorus-31?
Answers: 1

Chemistry, 23.06.2019 07:40
Did the detergents containing enzymes work better at removing stains than those containing no enzyme? why or why not?
Answers: 2
You know the right answer?
A weather balloon is inflated to a volume of 27.6 L at a pressure of 755 mmHg and a temperature of 2...
Questions




Mathematics, 23.08.2019 18:30

Mathematics, 23.08.2019 18:30


Mathematics, 23.08.2019 18:30

History, 23.08.2019 18:30

History, 23.08.2019 18:30

Mathematics, 23.08.2019 18:30





Physics, 23.08.2019 18:30




Mathematics, 23.08.2019 18:30

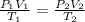
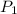 = initial pressure of gas = 755 mm Hg
= initial pressure of gas = 755 mm Hg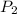 = final pressure of gas (at STP) = 385 mm Hg
= final pressure of gas (at STP) = 385 mm Hg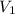 = initial volume of gas = 27.6 L
= initial volume of gas = 27.6 L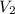 = final volume of gas = ?
= final volume of gas = ?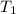 = initial temperature of gas =
= initial temperature of gas = 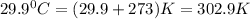
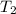 = final temperature of gas =
= final temperature of gas =