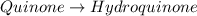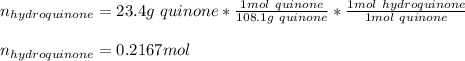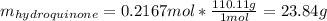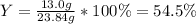Chemistry, 04.07.2020 01:01 allieb12334
Determine the theoretical maximum moles of hydroquinone, , that could be produced in this experiment. The reactant, quinone, is the limiting reagent. (To avoid introducing rounding errors on intermediate calculations, enter your answer to four significant figures.)
Reactant mass 23.4g
Product mass 13.0g
Reactant moles 0.2167 mol
Reactant mass 23.4g
Product mass 13.0g
Molar mass C 12.0 g/mol
Molar mass H 1.00 g/mol
Molar mass O 16.0 g/mol
Theoretical maximum moles of hydroquinone:

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
In which phase(s) do the molecules take the shape of the container?
Answers: 1



Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3
You know the right answer?
Determine the theoretical maximum moles of hydroquinone, , that could be produced in this experiment...
Questions


Mathematics, 07.10.2019 17:30

English, 07.10.2019 17:30


Advanced Placement (AP), 07.10.2019 17:30


Mathematics, 07.10.2019 17:30

History, 07.10.2019 17:30

Mathematics, 07.10.2019 17:30


Biology, 07.10.2019 17:30

History, 07.10.2019 17:30




English, 07.10.2019 17:30




Mathematics, 07.10.2019 17:30