Chemistry, 03.07.2020 22:01 andybiersack154
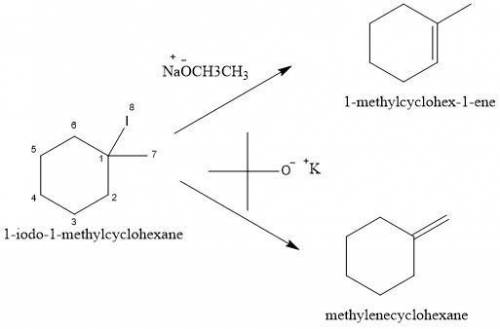
When 1-iodo-1-methylcyclohexane is treated with NaOCH2CH3 as the base, the more highly substituted alkene product predominates. When KOC(CH3)3 is used as the base, the less highly substituted alkene predominates. Give the structures of the two products and offer an explanation.

Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:00
Read these sentences from the text. near the equator, the tropics receive the most rain on a consistent basis. as a result, the fresh water falling into the ocean decrease the salinity of the surface water in that region. [. .] . . as the salt content of sea water increases, so does its density. what can you infer about how rain affects the density of surface water near the equator?
Answers: 1

Chemistry, 22.06.2019 09:20
What happened to the amount of carbon dioxide in the atmosphere from 2010–2017?
Answers: 1

Chemistry, 22.06.2019 22:30
Essay-alternative energy sources research sources of energy that are being developed. write a report of 350-400 words discussing the information you learned concerning the development of various energy sources and the impact that you think they will have on your life. include sources cited at the end of your report using the mla format. follow the rubric guidelines. note that wikipedia is not an appropriate resource for a research paper. worth 99
Answers: 3

Chemistry, 23.06.2019 02:00
What is the difference between a substance "getting wet" and "being dissolved" in a liquid at the particulate level?
Answers: 3
You know the right answer?
When 1-iodo-1-methylcyclohexane is treated with NaOCH2CH3 as the base, the more highly substituted a...
Questions

Mathematics, 08.02.2022 06:10

Health, 08.02.2022 06:10


Mathematics, 08.02.2022 06:10

Mathematics, 08.02.2022 06:10

Biology, 08.02.2022 06:10

Social Studies, 08.02.2022 06:10

Mathematics, 08.02.2022 06:10


English, 08.02.2022 06:20



Mathematics, 08.02.2022 06:20


Chemistry, 08.02.2022 06:20

English, 08.02.2022 06:20



Mathematics, 08.02.2022 06:20

English, 08.02.2022 06:20

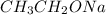 is a strong base but only has 2 carbons therefore we will have less steric hindrance in this base. So, the base can remove hydrogens that are bonded on carbons 1 or 6, therefore, we will have a more substituted alkene (1-methylcyclohex-1-ene).
is a strong base but only has 2 carbons therefore we will have less steric hindrance in this base. So, the base can remove hydrogens that are bonded on carbons 1 or 6, therefore, we will have a more substituted alkene (1-methylcyclohex-1-ene).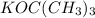 we have more steric hindrance. So, we can remove only the hydrogens from carbon 7 and we will produce a less substituted alkene (methylenecyclohexane).
we have more steric hindrance. So, we can remove only the hydrogens from carbon 7 and we will produce a less substituted alkene (methylenecyclohexane).