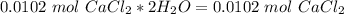Chemistry, 02.07.2020 03:01 Fangflora3
calculate how many moles of CaCl2•2H2O are present in 1.50 g of CaCl2•2H2O and then calculate how many moles of pure CaCl2 are present in the 1.50 g of CaCl2•2H2O.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 02:20
In a chemical reaction, the final amount of the products is determined by the a. universal gas law b. law of definite proportions c. air pressure d. temperature e. none of the above me
Answers: 2

Chemistry, 23.06.2019 03:00
In which of the following phases of matter do molecules have the highest amount of energy? a. liquid b. gel c. solid d. gas
Answers: 2
You know the right answer?
calculate how many moles of CaCl2•2H2O are present in 1.50 g of CaCl2•2H2O and then calculate how ma...
Questions


Health, 25.03.2021 01:00

Mathematics, 25.03.2021 01:00

Mathematics, 25.03.2021 01:00

Biology, 25.03.2021 01:00

Mathematics, 25.03.2021 01:00

Mathematics, 25.03.2021 01:00

Social Studies, 25.03.2021 01:00








History, 25.03.2021 01:00


Chemistry, 25.03.2021 01:00

Mathematics, 25.03.2021 01:00

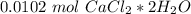
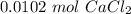
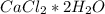 . For this, we have to know the atomic mass of each atom:
. For this, we have to know the atomic mass of each atom: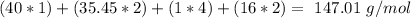
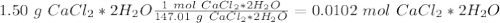
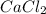 . Therefore, we have a 1:1 mol ratio . With this in mind, we will have the same number of moles for
. Therefore, we have a 1:1 mol ratio . With this in mind, we will have the same number of moles for