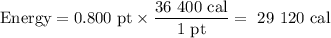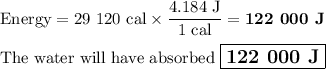Chemistry, 01.07.2020 15:01 kpruitt6359
Duncan knows that it takes 36400 cal of energy to heat a pint of water from room temperature to boiling. However, Duncan has prepared ramen noodles so many times he does not need to measure the water carefully. If he happens to heat 0.800 pint of room-temperature water, how many kilojoules of heat energy will have been absorbed by the water at the moment it begins to boil?

Answers: 3
Another question on Chemistry



Chemistry, 22.06.2019 05:30
Which of the following two events occur to create a sea breeze? select all that apply. warm air rises on the ocean and moves toward the land to cool warm air rises on land and moves toward the ocean to cool cool air moves from the ocean to be warmed by the land cool air moves from the land to be warmed by the ocean
Answers: 3

Chemistry, 22.06.2019 06:30
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2
You know the right answer?
Duncan knows that it takes 36400 cal of energy to heat a pint of water from room temperature to boil...
Questions

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

World Languages, 11.09.2020 16:01

Social Studies, 11.09.2020 16:01

Biology, 11.09.2020 16:01