Chemistry, 29.06.2020 01:01 xthom001ow7go3
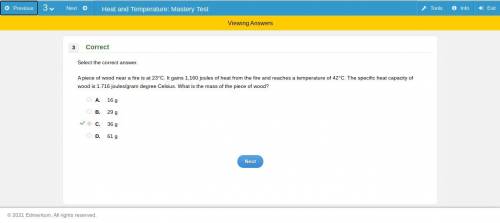
A piece of wood near a fire is at 23°C. It gains 1,160 joules of heat from the fire and reaches a temperature of 42°C. The specific heat capacity of
wood is 1.716 joules/gram degree Celsius. What is the mass of the piece of wood?
ОА. 16 g
OB. 29 g
ОC. 36 g
OD. 61 g

Answers: 3
Another question on Chemistry


Chemistry, 22.06.2019 07:30
In the particles are arranged in a regular, repeating pattern. a)a crystalline liquid b)a crystalline solid c)all gases d)all solids
Answers: 2

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1
You know the right answer?
A piece of wood near a fire is at 23°C. It gains 1,160 joules of heat from the fire and reaches a te...
Questions

Social Studies, 24.08.2020 06:01



Mathematics, 24.08.2020 06:01

Mathematics, 24.08.2020 06:01

Mathematics, 24.08.2020 06:01

Mathematics, 24.08.2020 06:01

Mathematics, 24.08.2020 06:01


Mathematics, 24.08.2020 06:01


Mathematics, 24.08.2020 06:01

Mathematics, 24.08.2020 06:01

English, 24.08.2020 06:01

Mathematics, 24.08.2020 06:01



Geography, 24.08.2020 06:01

Mathematics, 24.08.2020 06:01

Spanish, 24.08.2020 06:01