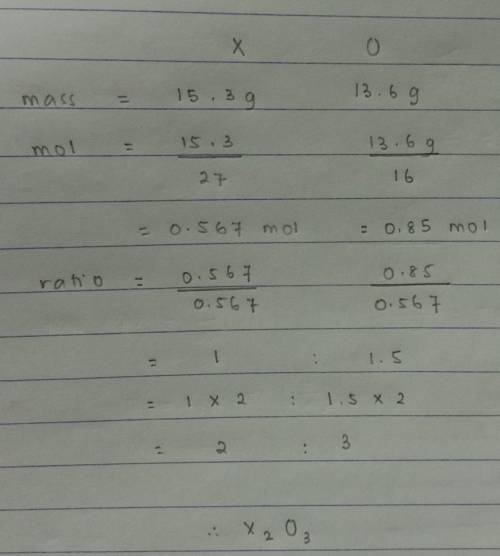Chemistry, 27.06.2020 15:01 Tariah5970
Quick answer plzAn oxide was prepared by combining 15.3g of an element “X” with 13.6g of oxygen. What is the simplest formula for the oxide? (At. Mass X = 27, O = 16)
A. XO3
B. X2O3
C. X2O5
D. XO

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 14:40
Choose an equation that represents an enzyme-catalyzed reaction. (a) enzyme + substrate → enzyme-substrate complex (b) enzyme + substrate ←−→ enzyme + products (c) enzyme + substrate ←−→ enzyme-substrate complex → enzyme + products (d) enzyme + substrate ←−→ enzyme-substrate complex → enzyme-substrate complex + products
Answers: 2

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Quick answer plzAn oxide was prepared by combining 15.3g of an element “X” with 13.6g of oxygen. Wha...
Questions



Mathematics, 20.05.2020 03:57

History, 20.05.2020 03:57






Mathematics, 20.05.2020 03:57

Mathematics, 20.05.2020 03:57

Mathematics, 20.05.2020 03:57

Mathematics, 20.05.2020 03:57

Biology, 20.05.2020 03:57

Mathematics, 20.05.2020 03:57



English, 20.05.2020 03:57

Arts, 20.05.2020 03:57

English, 20.05.2020 03:57