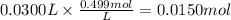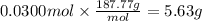Chemistry, 27.06.2020 05:01 viktoria1198zz
Aqueous solutions of copper (II) bromide and silver (1) acetate react to form solid
silver (1) bromide and aqueous copper (II) acetate according to the UNBALANCED
reaction below.
CuBr2 (aq) + AGCH3CO2 (aq)
-
AgBr (s) + Cu(CH3CO2)2 (aq)
How many grams of silver (1) bromide will form if 30.0 mL of 0.499 M copper (II)
bromide react with excess silver (1) acetate?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 13:00
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2

Chemistry, 22.06.2019 23:00
Which type of intermolecular attractions holds ammonia molecules together with other ammonia molecules?
Answers: 3

Chemistry, 23.06.2019 02:10
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2

Chemistry, 23.06.2019 07:00
In order for a high temperature boiler or steam engine to produce superheated water, or steam: the heat source must be greater than 100°c the water must be permitted to evaporate quickly the system must be sealed and become pressurized above atmospheric pressure the vapor pressure must be kept below 760 mm(hg)
Answers: 1
You know the right answer?
Aqueous solutions of copper (II) bromide and silver (1) acetate react to form solid
silver (1) brom...
Questions

English, 26.11.2019 05:31


Chemistry, 26.11.2019 05:31


Mathematics, 26.11.2019 05:31

English, 26.11.2019 05:31



Mathematics, 26.11.2019 05:31

Mathematics, 26.11.2019 05:31

Mathematics, 26.11.2019 05:31