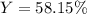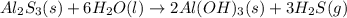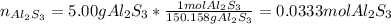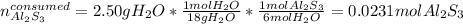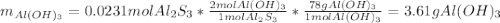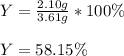Chemistry, 26.06.2020 15:01 rainbowboi
When 5.00 g of Al2S3 and 2.50 g of H2O are reacted according to the following reaction: Al2S3(s) + 6 H2O(l) → 2 Al(OH)3(s) + 3 H2S(g) 2.10 g were obtained. What is the percent yield of the reaction?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:30
Why are people not able to scuba dive in the deep part of the ocean
Answers: 2

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 18:40
What is the binding energy of a nucleus that has a mass defect of 5.81*10-^29 kg a 5.23*10-^12 j b 3.15* 10^12 j c 1.57*10-3 j d 9.44*10^20 j
Answers: 1

Chemistry, 23.06.2019 00:30
What is calcium oxide+diphosphorus pentoxide--> calcium phosphate balanced
Answers: 1
You know the right answer?
When 5.00 g of Al2S3 and 2.50 g of H2O are reacted according to the following reaction: Al2S3(s) + 6...
Questions


English, 18.11.2020 07:30


Mathematics, 18.11.2020 07:30

Social Studies, 18.11.2020 07:30

Mathematics, 18.11.2020 07:30



Health, 18.11.2020 07:30



SAT, 18.11.2020 07:30




Business, 18.11.2020 07:30