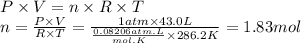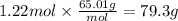The airbags that protect people in car crashes are inflated by the extremely rapid decomposition of sodium azide, which produces large volumes of nitrogen gas. 1. Write a balanced chemical equation, including physical state symbols, for the decomposition of solid sodium azide () into solid sodium and gaseous dinitrogen. 2. Suppose of dinitrogen gas are produced by this reaction, at a temperature of and pressure of exactly . Calculate the mass of sodium azide that must have reacted. Round your answer to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 18:00
Now consider the reaction when 45.0 g naoh have been added. what amount of naoh is this, and what amount of fecl3 can be consumed by it?
Answers: 3

Chemistry, 22.06.2019 03:00
Schrodinger and heisenberg developed an alternate theory about atomic nature that contradicted some of bohr's model of the atom. how do changes resulting from new technology and evidence affect the reputation of the atomic theory?
Answers: 1

Chemistry, 22.06.2019 19:00
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1

You know the right answer?
The airbags that protect people in car crashes are inflated by the extremely rapid decomposition of...
Questions





Mathematics, 12.11.2020 18:30

Arts, 12.11.2020 18:30