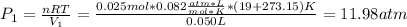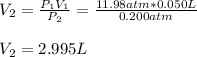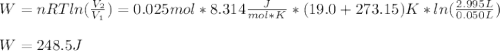A quantity of 0.0250 mol of a gas initially at 0.050 L and 19.0°C undergoes a constant-temperature expansion against a constant pressure of 0.200 atm. If the gas is allowed to expand unchecked until its pressure is equal to the external pressure, what would its final volume be before it stopped expanding, and what would be the work done by the gas?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
Match the name of the following compound: mgso4 · h2omagnesium sulfate monohydratemagnesium (ii) sulfate monohydratemagnesium (ii) sulfate hydratemagnesium sulfate hydrate
Answers: 1

Chemistry, 22.06.2019 12:50
The number at the end of an isotope’s name is the number.
Answers: 1

Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 1

Chemistry, 22.06.2019 18:30
The number of moles of a given mass of a substance can be found without knowing its molecular formula or molar mass. true false
Answers: 1
You know the right answer?
A quantity of 0.0250 mol of a gas initially at 0.050 L and 19.0°C undergoes a constant-temperature e...
Questions

Mathematics, 04.02.2020 10:00


Mathematics, 04.02.2020 10:00


Mathematics, 04.02.2020 10:00

History, 04.02.2020 10:00



History, 04.02.2020 10:00

Mathematics, 04.02.2020 10:00


Mathematics, 04.02.2020 10:00

English, 04.02.2020 10:00

English, 04.02.2020 10:00

Mathematics, 04.02.2020 10:00


English, 04.02.2020 10:00

Spanish, 04.02.2020 10:00

English, 04.02.2020 10:00

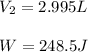
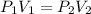
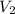 by firstly computing the initial pressure:
by firstly computing the initial pressure: