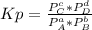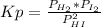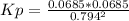Chemistry, 26.06.2020 15:01 wrightstephanie193
2. A reaction vessel is charged with hydrogen iodide, which partially decomposes to molecular hydrogen and iodine:2HI (g) H2(g) + I2(g)When the system comes to equilibrium at 425 °C, PHI = 0.794 atm, and PH2 = PI2 = 0.0685 atm. The value of Kp at this temperature is .

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
You are to give ampicillin with a recommended dose of 25mg/kg to a child with a mass of 29kg. if stock on hand is 250mg/capsule how many capsules should be given?
Answers: 1


Chemistry, 23.06.2019 00:00
How many atoms or molecules are there in a mole of a substance?
Answers: 1

Chemistry, 23.06.2019 04:20
The reaction below shows a system in equilibrium. how would a decrease in temperature affect this reaction? a. the rate of formation of the gases would increase. b. the equilibrium of the reaction would shift to the left. c. the equilibrium would shift to cause the gases to sublime into solids. d. the chemicals on the left would quickly form the chemical on the right.
Answers: 1
You know the right answer?
2. A reaction vessel is charged with hydrogen iodide, which partially decomposes to molecular hydrog...
Questions



Mathematics, 10.12.2020 21:10

Mathematics, 10.12.2020 21:10

Mathematics, 10.12.2020 21:10

Mathematics, 10.12.2020 21:10

Mathematics, 10.12.2020 21:10


Business, 10.12.2020 21:10

Mathematics, 10.12.2020 21:10


Mathematics, 10.12.2020 21:10

History, 10.12.2020 21:10

Biology, 10.12.2020 21:10

History, 10.12.2020 21:10

Mathematics, 10.12.2020 21:10




Geography, 10.12.2020 21:10