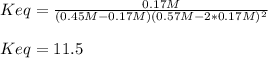Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 23.06.2019 01:00
Which description best characterization the motion of particles in a solid
Answers: 1

Chemistry, 23.06.2019 07:30
Achemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. the drug molecules bind the protein in a 1: 1 ratio to form a drug-protein complex. the protein concentration in aqueous solution at 25 ˚c is 1.74 x10-6 m . drug a is introduced into the protein solution at an initial concentration of 2.00 x10-6m. drug b is introduced into a separate, identical protein solution at an initial concentration of 2.00 x10-6m. at equilibrium, the drug a-protein solution has an a-protein complex concentration of 1.00 x10-6m, and the drug b solution has a b-protein complex concentration of 1.40 x10-6m.a. calculate the kc value for the a-protein binding reaction.b. calculate the kc value for the b-protein binding reaction.c. assuming that the drug that binds more strongly will be more effective, which drug is the better choice for further research?
Answers: 1

You know the right answer?
9. Given the following reaction:CO (g) + 2 H2(g) CH3OH (g)In an experiment, 0.45 mol of CO and 0.57...
Questions

English, 04.11.2020 15:50

Mathematics, 04.11.2020 15:50

Geography, 04.11.2020 15:50

History, 04.11.2020 15:50

Physics, 04.11.2020 15:50

Chemistry, 04.11.2020 15:50


History, 04.11.2020 15:50





Spanish, 04.11.2020 15:50


English, 04.11.2020 15:50

Mathematics, 04.11.2020 15:50

Mathematics, 04.11.2020 15:50

Mathematics, 04.11.2020 15:50

English, 04.11.2020 15:50

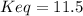
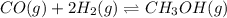
![Keq=\frac{[CH_3OH]}{[CO][H_2]^2}](/tpl/images/0694/1862/81c3a.png)
 due to the reaction extent we can write:
due to the reaction extent we can write:![Keq=\frac{x}{([CO]_0-x)([H_2]_0-2x)^2}](/tpl/images/0694/1862/e4583.png)
![[CO]_0=\frac{0.45mol}{1.00L}=0.45M\\](/tpl/images/0694/1862/3035d.png)
![[H_2]_0=\frac{0.57mol}{1.00L}=0.57M\\](/tpl/images/0694/1862/dc61d.png)
![[CO]_{eq}=\frac{0.28mol}{1.00L}=0.28M\\](/tpl/images/0694/1862/ecf3a.png)
![x=[CO]_0-[CO]_{eq}=0.45M-0.28M=0.17M](/tpl/images/0694/1862/8aaf6.png)