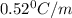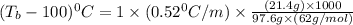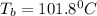Chemistry, 25.06.2020 03:01 michellegregg10
An ethylene glycol solution contains 21.4 g of ethylene glycol (C2H6O2) in 97.6 mL of water. (Assume a density of 1.00 g/mL for water.) Determine the freezing point and boiling point of the solution. (Assume a density of 1.00 g/ mL for water.)

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Match each object to its description: a. coma of a comet b. comet tail c. oort cloud haze surrounding a nucleus created by solar wind. hypothetical sphere around the solar system
Answers: 1

Chemistry, 22.06.2019 06:00
How many atoms of mg are present in 97.22 grams of mg? 6.022 × 1023 2.408 × 1024 4.818 × 1024 5.855 × 1025
Answers: 3


Chemistry, 22.06.2019 14:00
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2
You know the right answer?
An ethylene glycol solution contains 21.4 g of ethylene glycol (C2H6O2) in 97.6 mL of water. (Assume...
Questions







Business, 10.07.2019 10:20

Mathematics, 10.07.2019 10:20



Mathematics, 10.07.2019 10:20

Mathematics, 10.07.2019 10:20

Physics, 10.07.2019 10:20

Physics, 10.07.2019 10:20

Business, 10.07.2019 10:20

History, 10.07.2019 10:20


Mathematics, 10.07.2019 10:30

Biology, 10.07.2019 10:30

Biology, 10.07.2019 10:30

 and
and 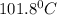 respectively.
respectively.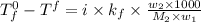
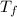 = freezing point of solution = ?
= freezing point of solution = ?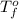 = freezing point of water =
= freezing point of water = 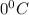
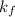 = freezing point constant of water =
= freezing point constant of water = 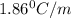
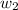 = mass of solute (ethylene glycol) = 21.4 g
= mass of solute (ethylene glycol) = 21.4 g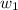 = mass of solvent (water) =
= mass of solvent (water) = 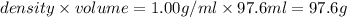
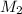 = molar mass of solute (ethylene glycol) = 62g/mol
= molar mass of solute (ethylene glycol) = 62g/mol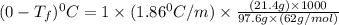
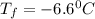
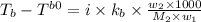
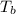 = boiling point of solution = ?
= boiling point of solution = ?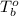 = boiling point of water =
= boiling point of water = 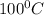
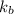 = boiling point constant of water =
= boiling point constant of water =