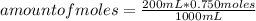Chemistry, 24.06.2020 23:01 berniceallonce22
Suppose you have 200.0 mL of a 0.750 M sodium hydroxide solution. How many moles of sodium hydroxide are in the solution?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:10
The covalent compound acetylene, which is the fuel of the oxyacetylene torch used by welders, has the molecular formula c2h2. the covalent compound benzene, a commercial solvent, has the molecular formula c6h6 each of these covalent compounds contains carbon and hydrogen atoms in a one-to-one ratio. would it be correct to write the chemical formulas of each as ch? explain.
Answers: 1


Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2

Chemistry, 23.06.2019 03:30
Ahelium balloon contains 16.9 l of helium at stp. how many atoms of helium are in the balloon
Answers: 1
You know the right answer?
Suppose you have 200.0 mL of a 0.750 M sodium hydroxide solution. How many
moles of sodium hydroxid...
Questions


Mathematics, 24.06.2019 22:30

Mathematics, 24.06.2019 22:30




History, 24.06.2019 22:30


Biology, 24.06.2019 22:30

Mathematics, 24.06.2019 22:30

History, 24.06.2019 22:30

History, 24.06.2019 22:30


Health, 24.06.2019 22:30


Mathematics, 24.06.2019 22:30



History, 24.06.2019 22:30

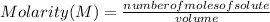
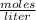 ).
).