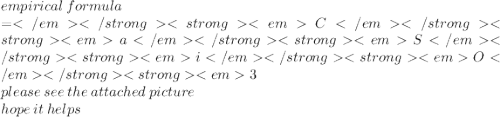Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

Chemistry, 22.06.2019 16:30
Asample of freon gas has a volume of 2.23 liters, a pressure of 4.85 kpa, and a temperature of -1.36°c. calculate the volume at a pressure of 1.38 kpa and a temperature of 5.5°c. (show work)
Answers: 1

Chemistry, 22.06.2019 16:30
How many moles of sulfuric acid (h2so4) are needed to react completely with 6.8 moles of lithium hydroxide (lioh)? 2lioh + h2so4 → li2so4 + 2h2o a. 3.4 mol h2so4b. 6.8 mol h2so4 c. 10.2 mol h2so4 d. 13.6 mol h2so4
Answers: 3
You know the right answer?
A compound contains 34.5% calcium, 24.1% silicon and 41.4% oxygen by mass. What is its empirical for...
Questions


Mathematics, 03.10.2019 06:30






Mathematics, 03.10.2019 06:30

French, 03.10.2019 06:30


Mathematics, 03.10.2019 06:30

Social Studies, 03.10.2019 06:30

Biology, 03.10.2019 06:30


Social Studies, 03.10.2019 06:30




Health, 03.10.2019 06:30