
Chemistry, 21.06.2020 20:57 aghamuzahirali4392
In this reaction, how many grams of O2 are required to completely react with 110 grams of Al

Answers: 1
Another question on Chemistry

Chemistry, 23.06.2019 00:30
What is the percent by mass of magnesium sulfate in mgso4.7h2o
Answers: 3


Chemistry, 23.06.2019 03:30
Ineed pls urgent 1-20 in order and fully detail step my step.
Answers: 1

Chemistry, 23.06.2019 07:00
Determine the length of the object shown. 97.8 mm 97.80 mm 97 mm 98 mm
Answers: 1
You know the right answer?
In this reaction, how many grams of O2 are required to completely react with 110 grams of Al...
Questions

Mathematics, 19.09.2019 22:30







Chemistry, 19.09.2019 22:30



Mathematics, 19.09.2019 22:30









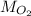 )Calculate molar mass of aluminum (
)Calculate molar mass of aluminum (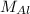 )Use
)Use 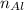 )With
)With 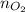 )Use
)Use 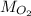 to find the mass of oxygen (
to find the mass of oxygen (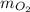 )
)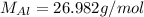
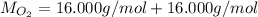
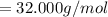
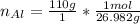 Multiply mass by molar mass to find moles.
Multiply mass by molar mass to find moles.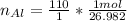 The units "g" cancel out.
The units "g" cancel out.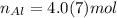 Keep one extra significant figure. (110 has 2 sig. figs.)
Keep one extra significant figure. (110 has 2 sig. figs.)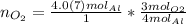 Multiply moles of aluminum by the mole ratio.
Multiply moles of aluminum by the mole ratio.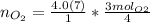 "molAl" units cancel out.
"molAl" units cancel out.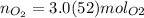 Keep two sig. figs. when the first is a "5"
Keep two sig. figs. when the first is a "5"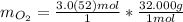 Multiply moles by molar mass.
Multiply moles by molar mass.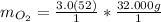 The "mol" unit cancels out.
The "mol" unit cancels out.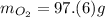 Keep one sig. fig. to round. "6" rounds up.
Keep one sig. fig. to round. "6" rounds up.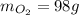 <= Final answer
<= Final answer

