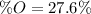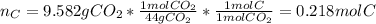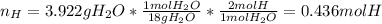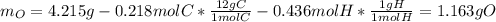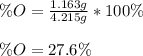Chemistry, 20.06.2020 17:57 collin0123
A 4.215 g sample of a compound containing only carbon, hydrogen, and oxygen is burned in an excess of oxygen gas, producing 9.582 g CO2 and 3.922 g H2O. What percent by mass of oxygen is contained in the original sample?

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 00:00
The pressure inside a hydrogen-filled container was 2.10 atm at 21 ? c. what would the pressure be if the container was heated to 92 ? c ?
Answers: 2

Chemistry, 22.06.2019 07:00
Which atom or ion is the largest? a. k b. k+ c. ca d. ca2+ e. li
Answers: 1

Chemistry, 22.06.2019 09:30
Which element is the least metallic between cadmium, silver, zinc, or iron?
Answers: 1

Chemistry, 22.06.2019 14:30
How does a noncompetitive inhibitor reduce an enzyme’s activity?
Answers: 1
You know the right answer?
A 4.215 g sample of a compound containing only carbon, hydrogen, and oxygen is burned in an excess o...
Questions

English, 01.12.2020 07:20

History, 01.12.2020 07:20


History, 01.12.2020 07:20





Biology, 01.12.2020 07:20



Mathematics, 01.12.2020 07:20


Mathematics, 01.12.2020 07:20






Mathematics, 01.12.2020 07:20