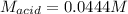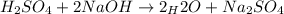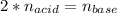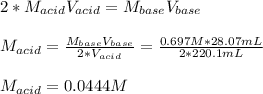Suppose you are titrating a sulfuric acid solution of unknown concentration with a sodium hydroxide solution according to the equation H 2 S O 4 + 2 N a O H ⟶ 2 H 2 O + N a 2 S O 4 If you require 28.07 mL of 0.697 M NaOH solution to titrate 220.1 mL of H 2 SO 4 solution, what is the concentration of the H 2 SO 4 solution? Type

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 06:30
Design techniques and materials that reduce the negative environmental impact of a structure are referred to as
Answers: 2

Chemistry, 22.06.2019 07:00
How heavy is thanos? a) 3000 lbs b) all of it c) the price of tea in china d) heavy enough
Answers: 2

Chemistry, 22.06.2019 15:30
The reactions of photosynthesis occur in the of plant cell? a.mitochondria. b. lysosomes. c. chloroplasts. d. chlorophyll
Answers: 1
You know the right answer?
Suppose you are titrating a sulfuric acid solution of unknown concentration with a sodium hydroxide...
Questions


English, 16.06.2021 02:00

Biology, 16.06.2021 02:00

Mathematics, 16.06.2021 02:00




Mathematics, 16.06.2021 02:00


Mathematics, 16.06.2021 02:00

Mathematics, 16.06.2021 02:00

Mathematics, 16.06.2021 02:00



History, 16.06.2021 02:00


Mathematics, 16.06.2021 02:00


Mathematics, 16.06.2021 02:00