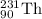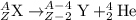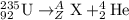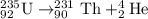Chemistry, 20.06.2020 04:57 akatherine1738
Consider the nuclear equation below. Superscript 235 subscript 92 upper U right arrow superscript 4 subscript 2 upper H e. What is the nuclide symbol of X? Superscript 231 subscript 94 upper P u. Superscript 235 subscript 90 upper T h. Superscript 239 subscript 94 upper P u. Superscript 231 subscript 90 upper T h. the nuclear equation below.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:00
Why do sodium and neon have vastly different chemical and physical properties despite having similar atomic masses?
Answers: 2

Chemistry, 22.06.2019 06:00
Calculate the mass of silver needed to react with chlorine to produce 126g if silver chloride?
Answers: 3

Chemistry, 22.06.2019 11:30
Which statement best describes the flow of energy in this scenario
Answers: 1

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
You know the right answer?
Consider the nuclear equation below. Superscript 235 subscript 92 upper U right arrow superscript 4...
Questions



Mathematics, 29.07.2021 01:00


History, 29.07.2021 01:00





Mathematics, 29.07.2021 01:00

Mathematics, 29.07.2021 01:00


History, 29.07.2021 01:00




Mathematics, 29.07.2021 01:00

Mathematics, 29.07.2021 01:00