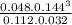Problem PageQuestionSteam reforming of methane ( ) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. An industrial chemist studying this reaction fills a tank with of methane gas and of water vapor, and when the mixture has come to equilibrium measures the amount of hydrogen gas to be .Calculate the concentration equilibrium constant for the steam reforming of methane at the final temperature of the mixture. Round your answer to significant digits.

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 23:00
50 pts plz what is the physical state of matter of baking soda.
Answers: 1

Chemistry, 22.06.2019 11:00
When hydrochloric acid reacts with potassium hydroxide solution, the following reaction occurs. hcl (aq) + koh (aq) h2o (l) + kcl (aq) the reaction gives off heat energy, so it is an reaction.
Answers: 1


Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2
You know the right answer?
Problem PageQuestionSteam reforming of methane ( ) produces "synthesis gas," a mixture of carbon mon...
Questions


History, 10.12.2019 20:31


Chemistry, 10.12.2019 20:31

Biology, 10.12.2019 20:31



Physics, 10.12.2019 20:31

Chemistry, 10.12.2019 20:31




Mathematics, 10.12.2019 20:31




History, 10.12.2019 20:31


Biology, 10.12.2019 20:31

History, 10.12.2019 20:31

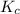 = 2.
= 2.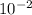
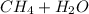 ⇒
⇒ 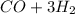
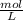 ) of each molecule of the reaction.
) of each molecule of the reaction.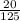 = 0.16M
= 0.16M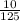 = 0.08M
= 0.08M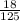 = 0.144M
= 0.144M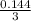 = 0.048M
= 0.048M![K_{c} = \frac{[CO][H_{2}]^{3} }{[CH_{4}][H_{2}O ] }](/tpl/images/0689/6943/3ac66.png)