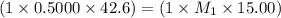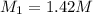Chemistry, 18.06.2020 15:57 umezinwachukwuebuka1
Ammonia (NH3) is a weak base that reacts with a strong acid to form the ammonium ion, NH4 . In the titration of 15.00 mL of an ammonia cleaner with 0.5000 M HCl, 42.6 mL of the titrant was required to reach the endpoint. What is the concentration of the NH3 in solution

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 2

Chemistry, 23.06.2019 00:30
The molecular weight of carbon dioxide, co2, is 44.00 amu, and the molecular weight of nitrous dioxide, no2, is 46.01 amu, so no2 diffuses co2
Answers: 2
You know the right answer?
Ammonia (NH3) is a weak base that reacts with a strong acid to form the ammonium ion, NH4 . In the t...
Questions


Mathematics, 10.12.2020 01:00

Mathematics, 10.12.2020 01:00

French, 10.12.2020 01:00






English, 10.12.2020 01:00


History, 10.12.2020 01:00

History, 10.12.2020 01:00



Mathematics, 10.12.2020 01:00

Health, 10.12.2020 01:00

Mathematics, 10.12.2020 01:00


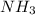 in solution is 1.42 M
in solution is 1.42 M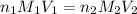
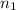 = basicity of
= basicity of 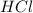 = 1
= 1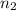 = acidity of
= acidity of 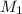 = concentration of
= concentration of 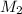 = concentration of
= concentration of 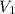 = volume of
= volume of 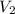 = volume of
= volume of