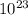Answers: 3
Another question on Chemistry

Chemistry, 21.06.2019 21:10
Identify one disadvantage to each of the following models of electron configuration: dot structures arrow and line diagrams written electron configurations type in your answer below.
Answers: 1

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 12:00
Which of the following is an example of physical change not a chemical change? a) a log gives off heat and light as it burns. b) a tree stores energy from the sun in its fruit. c) a penny lost in the grass slowly changes color. d) a water pipe freezes and cracks on a cold night.
Answers: 2

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1
You know the right answer?
How many atoms of carbon are in 4.00 moles of C6H12O6?...
Questions

Mathematics, 25.08.2019 21:10

Mathematics, 25.08.2019 21:10

Mathematics, 25.08.2019 21:10


Biology, 25.08.2019 21:10

Biology, 25.08.2019 21:10

English, 25.08.2019 21:10

Mathematics, 25.08.2019 21:10

Mathematics, 25.08.2019 21:10


Chemistry, 25.08.2019 21:10


Mathematics, 25.08.2019 21:10

English, 25.08.2019 21:10


Mathematics, 25.08.2019 21:10




Chemistry, 25.08.2019 21:10