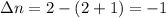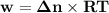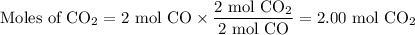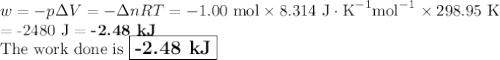Chemistry, 16.06.2020 02:57 estrellaalcantar16
Consider that you have a balloon containing 2.00 moles of CO and 1.00 mole of O2 which is in a room that has a temperature of 25.8oC and a pressure of 1.00 atm. Then, the following reaction occurs inside the balloon to completion: 2 CO(g) + O2(g) -> 2 CO2(g). Calculate the change in work due to the reaction occurring inside the balloon.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1


Chemistry, 22.06.2019 17:00
The arrangement of particles is most ordered in a sample of
Answers: 1

Chemistry, 22.06.2019 17:30
Take a look at this dandelion. the yellow flower on the right is pollinated and the seeds on the left are transported by
Answers: 2
You know the right answer?
Consider that you have a balloon containing 2.00 moles of CO and 1.00 mole of O2 which is in a room...
Questions









Mathematics, 29.01.2020 04:49




Mathematics, 29.01.2020 04:49

Chemistry, 29.01.2020 04:49


Law, 29.01.2020 04:50



Mathematics, 29.01.2020 04:50

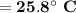
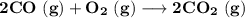
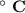 to
to 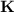 :
: 
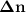 value:
value: