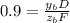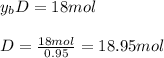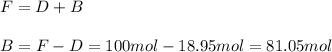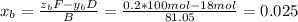Chemistry, 15.06.2020 09:57 ijeomaokoro2264
A mixture containing 20 mole % butane, 35 mole % pentane and rest
hexane, is to be separated by fractional distillation into a distillate
containing 95 mole % butane, 4 mole % pentane and rest hexane and
a bottom product. The distillate is expected to contain 90% of the
butane in the feed. Calculate the composition of the bottom product.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:30
In numbering carbon atoms in the parent chain of a hydrocarbon, why would you number from right to left, rather than left to right
Answers: 1

Chemistry, 22.06.2019 05:00
Agas can holds 2.0 gal of gasoline. what is this quantity in cubic centimeters?
Answers: 2

Chemistry, 22.06.2019 16:30
For the reaction shown, calculate how many moles of no2 form when each of the following completely reacts. 2n2o5(g)→4no2(g)+o2(g) part a 1.0 mol n2o5 express your answer using two significant figures. nothing mol m o l request answer part b 5.4 mol n2o5 express your answer using two significant figures.
Answers: 2

Chemistry, 22.06.2019 19:00
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1
You know the right answer?
A mixture containing 20 mole % butane, 35 mole % pentane and rest
hexane, is to be separated by fra...
Questions

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

Mathematics, 16.09.2020 08:01

History, 16.09.2020 08:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

Mathematics, 16.09.2020 09:01

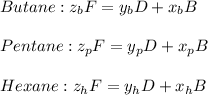
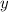 accounts for the fractions at the outlet distillate and
accounts for the fractions at the outlet distillate and  for the fractions at the outlet bottoms. Moreover, with the 90 % recovery of butane, we can write:
for the fractions at the outlet bottoms. Moreover, with the 90 % recovery of butane, we can write: