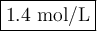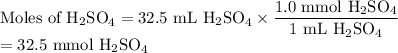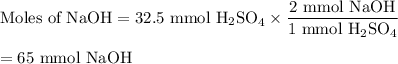Chemistry, 15.06.2020 01:57 gigimasters71p7tc6l
In a titration 32.5 mL of 1.0 M sulfuric acid is required to neutralize 45.0 mL of sodium hydroxide. What is the concentration of the base?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2


Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3

Chemistry, 23.06.2019 06:40
How many joules of heat are required to raise thetemperature of 750 g of water from 11.0 °c to 19.0 °c?
Answers: 1
You know the right answer?
In a titration 32.5 mL of 1.0 M sulfuric acid is required to neutralize 45.0 mL of sodium hydroxide....
Questions

Mathematics, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31


Chemistry, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31


Mathematics, 21.11.2019 08:31

Business, 21.11.2019 08:31


Mathematics, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31


Mathematics, 21.11.2019 08:31

Mathematics, 21.11.2019 08:31



English, 21.11.2019 08:31