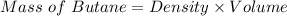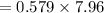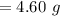Be sure to answer all parts. Butane gas is compressed and used as a liquid fuel in disposable cigarette lighters and lightweight camping stoves. Suppose a lighter contains 7.96 mL of butane (d = 0.579 g/mL). (a) How many grams of oxygen are needed to burn the butane completely?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3

Chemistry, 22.06.2019 08:30
What method(s) do plants use to obtain nitrogen? select all that apply. absorb it from the atmosphere use bacteria to convert nitrogen to usable form obtain usable nitrogen compounds from the soil absorb nitrogen from water taken in at the roots
Answers: 3

Chemistry, 22.06.2019 21:30
Liquid ammonia is produced at high temperatures and under great pressure in a tank by passing a mixture of nitrogen gas and hydrogen gas over an iron catalyst. the reaction is represented by the following equation. n2(g) + 3h2(g) → 2nh3(g) changing all but one experimental condition will affect the amount of ammonia produced. that condition is a) increasing the concentration of both reactants b) changing the temperature within the tank c) decreasing the pressure within the tank. d) increasing only the amount of nitrogen present.
Answers: 1

Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2
You know the right answer?
Be sure to answer all parts. Butane gas is compressed and used as a liquid fuel in disposable cigare...
Questions


History, 25.01.2020 20:31

Spanish, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

Biology, 25.01.2020 20:31




Mathematics, 25.01.2020 20:31


Health, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31



Biology, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31

Mathematics, 25.01.2020 20:31