Answer :
(1) 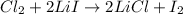
(2) 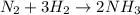
(3) 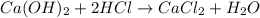
(4) 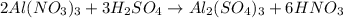
(5) 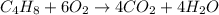
(6) 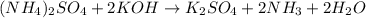
(7) 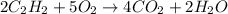
(8) 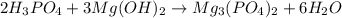
(9) 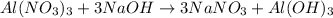
(10) 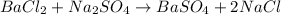
Explanation :
Balanced chemical reaction : It is defined as the reaction in which the number of atoms of individual elements present on reactant side must be equal to the product side.
Part 1 :
The unbalanced chemical reaction is,
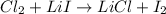
In order to balance the chemical equation, the coefficient '2' put before the 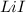 and
and 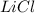 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
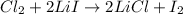
Part 2 :
The unbalanced chemical reaction is,
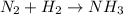
In order to balance the chemical equation, the coefficient '3' put before the 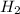 and the coefficient '2' put before the
and the coefficient '2' put before the 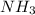 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
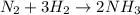
Part 3 :
The unbalanced chemical reaction is,
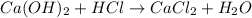
In order to balance the chemical equation, the coefficient '2' put before the 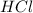 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
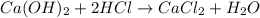
Part 4 :
The unbalanced chemical reaction is,
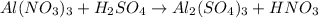
In order to balance the chemical equation, the coefficient '2' put before the 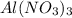 , the coefficient '3' put before the
, the coefficient '3' put before the 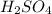 and the coefficient '6' put before the
and the coefficient '6' put before the 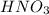 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
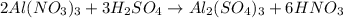
Part 5 :
The unbalanced chemical reaction is,
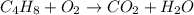
In order to balance the chemical equation, the coefficient '6' put before the 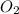 , the coefficient '4' put before the
, the coefficient '4' put before the 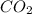 and the coefficient '4' put before the
and the coefficient '4' put before the 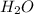 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
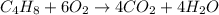
Part 6 :
The unbalanced chemical reaction is,
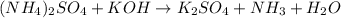
In order to balance the chemical equation, the coefficient '2' put before the 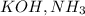 and
and 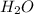 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
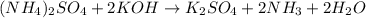
Part 7 :
The unbalanced chemical reaction is,
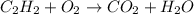
In order to balance the chemical equation,the coefficient '2' put before the 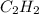 , the coefficient '5' put before the
, the coefficient '5' put before the 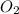 , the coefficient '4' put before the
, the coefficient '4' put before the 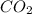 and the coefficient '2' put before the
and the coefficient '2' put before the 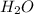 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
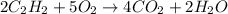
Part 8 :
The unbalanced chemical reaction is,
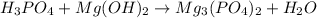
In order to balance the chemical equation,the coefficient '2' put before the 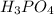 , the coefficient '3' put before the
, the coefficient '3' put before the 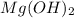 and the coefficient '6' put before the
and the coefficient '6' put before the 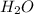 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
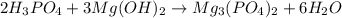
Part 9 :
The unbalanced chemical reaction is,
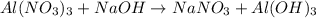
In order to balance the chemical equation,the coefficient '3' put before the 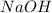 and the coefficient '3' put before the
and the coefficient '3' put before the 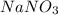 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
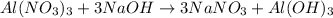
Part 10 :
The unbalanced chemical reaction is,
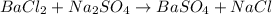
In order to balance the chemical equation,the coefficient '2' put before the 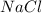 then we get the balanced chemical equation.
then we get the balanced chemical equation.
The balanced chemical reaction will be,
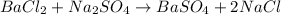


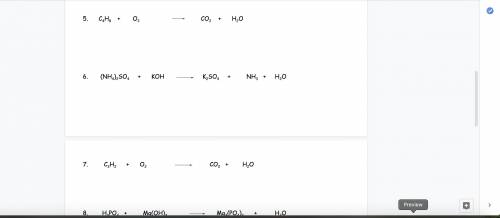



























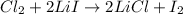
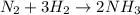
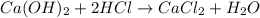
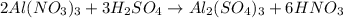
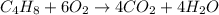
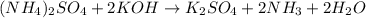
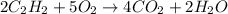
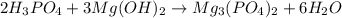
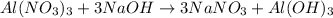
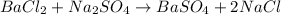
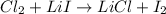
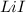 and
and 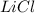 then we get the balanced chemical equation.
then we get the balanced chemical equation.
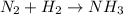
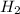 and the coefficient '2' put before the
and the coefficient '2' put before the 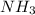 then we get the balanced chemical equation.
then we get the balanced chemical equation.
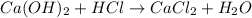
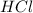 then we get the balanced chemical equation.
then we get the balanced chemical equation.
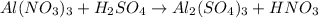
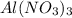 , the coefficient '3' put before the
, the coefficient '3' put before the 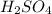 and the coefficient '6' put before the
and the coefficient '6' put before the 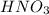 then we get the balanced chemical equation.
then we get the balanced chemical equation.
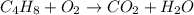
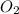 , the coefficient '4' put before the
, the coefficient '4' put before the 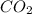 and the coefficient '4' put before the
and the coefficient '4' put before the 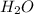 then we get the balanced chemical equation.
then we get the balanced chemical equation.
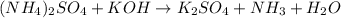
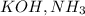 and
and 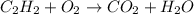
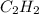 , the coefficient '5' put before the
, the coefficient '5' put before the 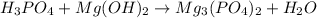
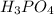 , the coefficient '3' put before the
, the coefficient '3' put before the 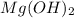 and the coefficient '6' put before the
and the coefficient '6' put before the 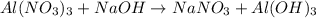
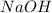 and the coefficient '3' put before the
and the coefficient '3' put before the 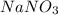 then we get the balanced chemical equation.
then we get the balanced chemical equation.
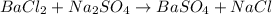
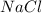 then we get the balanced chemical equation.
then we get the balanced chemical equation.


