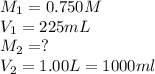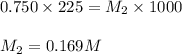Chemistry, 13.06.2020 18:57 MathChic68
To 225ml of a 0.750M solution of KL a student adds enough water to make 1.00L of a more dilute KL solution. What is the morality of the new solution? plz show work

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Mitosis is a type of cell division that produces cells that are identical to the parent cell. meiosis is a different type of cell division that produces cells that carry have a genetic material of the parent cell. based on the information provided how do the purpose of mitosis and meiosis differ
Answers: 3

Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

Chemistry, 22.06.2019 11:00
Iron (3) oxide will decompose in the presence of hydrogen gas and heater to produced iron and digydrogen monoxide white a balanced chemical equation
Answers: 1

Chemistry, 22.06.2019 18:30
Read the claim. breakfast is an important meal. it jump starts the body’s process of using calories to break down food. appetite can decrease with age, but going too long without eating causes metabolism to slow down. current research shows that incorporating legumes such as lentils and chickpeas into meals boosts metabolism for twenty-four hours. who might benefit from this claim? people who have a fast metabolism stores that sell exercise equipment people who take vitamin supplements grocery stores that sell legumes
Answers: 1
You know the right answer?
To 225ml of a 0.750M solution of KL a student adds enough water to make 1.00L of a more dilute KL so...
Questions



Mathematics, 05.04.2020 20:23

Mathematics, 05.04.2020 20:23

History, 05.04.2020 20:23



Mathematics, 05.04.2020 20:23

History, 05.04.2020 20:23


Mathematics, 05.04.2020 20:23


Mathematics, 05.04.2020 20:23




Mathematics, 05.04.2020 20:23


Chemistry, 05.04.2020 20:24

Mathematics, 05.04.2020 20:24

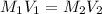
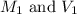 are the molarity and volume of concentrated solution
are the molarity and volume of concentrated solution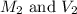 are the molarity and volume of diluted solution
are the molarity and volume of diluted solution